Producing "green hydrogen" via water electrolysis powered by renewable energy sources such as wind and solar is a critical pathway for consuming green electricity, decarbonizing heavy industry, and achieving carbon neutrality goals. Currently, alkaline water electrolysis stands as the mainstream technology for green hydrogen production due to its technological maturity and cost-effectiveness. However, coupling this technology with fluctuating renewable energy sources presents a significant challenge: frequent start-stop cycles trigger a severe "reverse current" phenomenon. This causes cathode oxidation and anode reduction, leading to electrode corrosion and interface delamination, which severely limits electrolysis efficiency and equipment lifespan. This dynamic stability issue has become a core bottleneck restricting the large-scale, high-efficiency coupling of alkaline electrolyzers with renewable energy.
Recently, a research team from the Industrial Deep Decarbonisation Research Center at Tsinghua University’s Institute for Carbon Neutrality and the Department of Chemical Engineering at Tsinghua University, led by Professor Zhang Qiang and Associate Researcher Tang Cheng, has achieved a major breakthrough in key materials for hydrogen production from fluctuating new energy sources. The team innovatively proposed a "Gradient Heterointerface Engineering" strategy, successfully developing novel electrode materials with superior resistance to reverse current and industrial-grade electrolysis performance. This achievement offers a groundbreaking material solution to overcome the critical bottlenecks of intermittent renewable energy electrolysis, holding great promise for driving the high-quality development of the green hydrogen industry.

Fig. 1. (a) Mechanism of "reverse current" generation after alkaline electrolyzer shutdown; (b) Schematic illustration of catalyst layer detachment and electrode corrosion during shutdown; (c) Schematic of the "Gradient Heterointerface Engineering" strategy and the resulting improvement in electrode stability.
Distinct from traditional approaches focusing on material composition and electronic structure optimization, the research team shifted their focus to the precise regulation of interfacial crystallographic structures. Using a thermal injection process, they grew a Ni3S2 catalytic layer with a Ni/Ni3S2 gradient heterointerface transition layer directly onto a commercial nickel mesh substrate. This unique structure creates a "seamless interface" that effectively alleviates lattice mismatch and stress between heterogeneous materials, promotes charge redistribution, and fundamentally enhances mechanical and electrochemical stability under drastic potential reversals and high current densities.
Under industrial testing conditions (80°C, 30 wt% KOH), the new electrode not only met the U.S. Department of Energy’s 2026 activity target for alkaline electrolysis (1000 mA cm⁻² @ 1.79 V) but also demonstrated zero performance degradation after 3,600 rigorous start-stop cycles. These results highlight its immense potential for real-world industrial application.
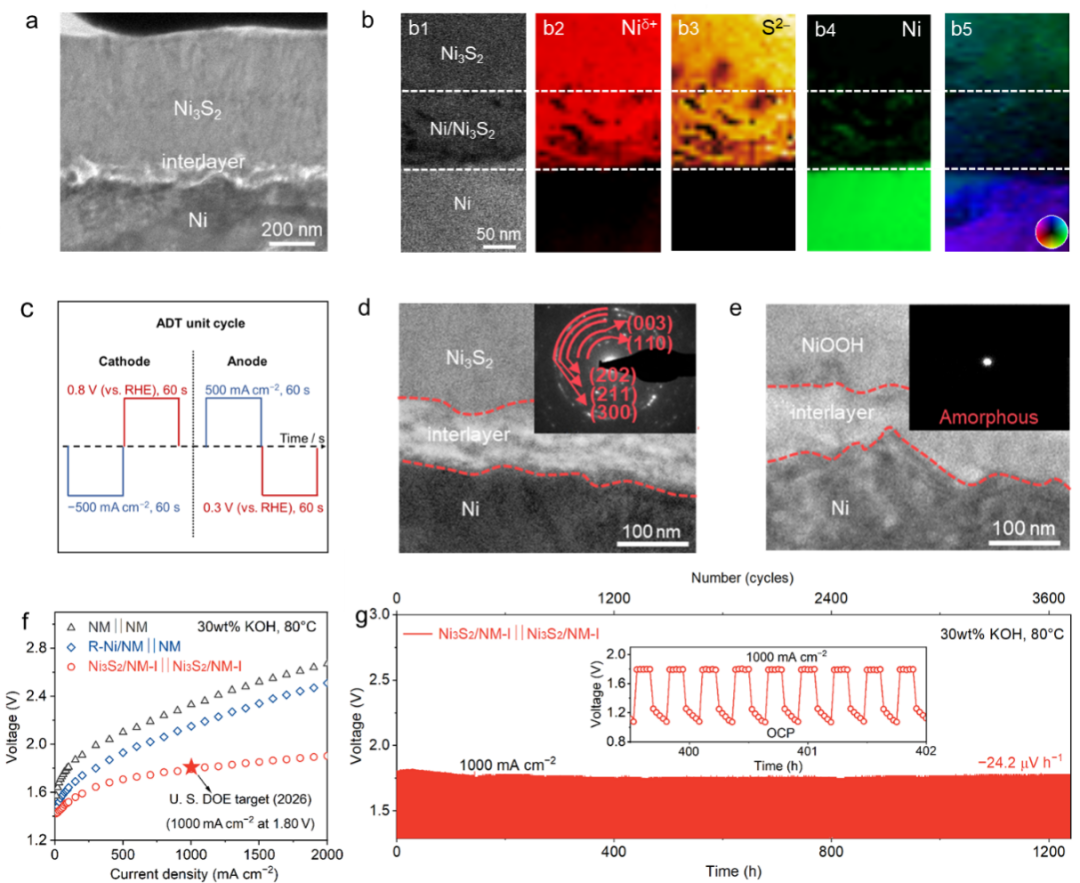
Fig. 2. (a, b) Characterization of the "substrate-transition layer-catalyst layer" interface; (c) Accelerated aging experiments simulating reverse current; Interface characterization of the electrode after 4,000 cycles of accelerated aging testing as (d) cathode and (e) anode; (f) Performance of the electrode under industrial conditions; (g) Start-stop stability test of the electrode under industrial conditions.
The research findings were published online on December 11 in the Journal of the American Chemical Society (JACS) under the title "Heterointerface-Enabled Anti-Reverse-Current Electrodes for Alkaline Water Electrolyzers at 1000 mA cm⁻²."
Professor Zhang Qiang and Associate Researcher Tang Cheng from Tsinghua University serve as the co-corresponding authors. The co-first authors are Dr. He Wenjun (Postdoctoral Fellow, Department of Chemical Engineering, Tsinghua University), Dr. Wang Yueshuai (Postdoctoral Fellow, School of Materials Science and Engineering, Beijing University of Technology), and Zhao Yilong (Master's student, School of Electrical and Electronic Engineering, Harbin University of Science and Technology).
This study was supported by the National Key R&D Program of China, the National Natural Science Foundation of China, the Huaneng Group Science and Technology Research Project, and the Tsinghua University Independent Research Plan.
 Latest recommendations
Latest recommendations


